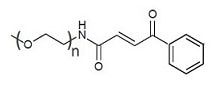
Description
mPEG-Carbonylacrylic (mPEG-CA) are for chemoselective cysteine bioconjugation. Carbonylacrylic PEG reagents undergo rapid thiol Michael-addition under biocompatible conditions in stoichiometric amounts. Importantly, the conjugates formed between Carbonylacrylic PEG and thiol/cysteine-containing molecules are resistant to degradation in physiological conditions. The typical maleimide-thiol reaction results in thiosuccinimide formation which is reversible, with PEG-maleimide elimination occurring slowly under biologically relevant conditions. The reversed reaction leads to the breakage of the maleimide-thiol linkage and the loss of PEG from the thiol/cysteine-containing molecules. If the maleimide-thiol chemistry does not work for your application, you may consider Carbonylacrylic PEG reagents as an improved alternative to PEG-maleimide.
Properties
Molecular weight: MW of PEG was measured by MALDI-MS or GPC. PDI (polydispersity index) of our linear PEG is 1.02-1.05 with very narrow MW distribution. The number of repeating ethylene oxide units (CH2CH2O) or the degree of polymerization is calculated dividing the PEG MW by 44 (44 is the molecular mass of one repeating unit).
Solubility: Soluble in water and aqueous buffer, chloroform, methylene chloride, DMF, DMSO, and less soluble in alcohol, toluene. Not soluble in ether.
Density: PEG density is approximately 1.125 g/mL
Physical form: PEG products generally appear as white or off-white powder, and for very low MW linear PEG such as MW 1k or less, it may appear as wax-like, semi-solid material due to the low MW and the type of functional groups.
Storage condition: PEG product shall be stored in the original form as received in a freezer at -20C or lower for long term storage. Stock solution of PEG reagents that do not contain oxygen or moisture sensitive functional groups may be temporarily stored in a refrigerator or ambient temperature for multiple days. Stock solution should avoid repeated freeze-and-thaw cycles. See Documents section for detailed storage and handling conditions.
References
1. Stoichiometric and irreversible cysteine-selective protein modification using carbonylacrylic reagent, Nature Communications | 7:13128 | DOI: 10.1038/ncomms13128, Text.
2. Tunable degradation of maleimide-thiol adducts in reducing environments. Bioconjug. Chem. 22, 1946–1953 (2011). Text.
3. Tunable reagents for multi-functional bioconjugation: reversible or permanent chemical modification of proteins and peptides by control of maleimide hydrolysis. Chem. Commun. (Camb.) 47, 5452–5454 (2011). Text.