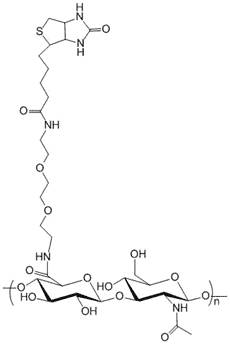
Description
Hyaluronic Acid is labeled with biotin through a long chain spacer. The long chain spacer is about 30 angstroms (3 nanometers). The loner linker provides easier access to the binding pocket of streptavidin by the biotin molecule in case of sterically hindered molecular interaction situations. Purity: >95% lyophilized powder. Standard degree of substitution (DoS): 1 mol % substitution and at least one biotin molecule per HA polymer. Synonyms: HA, hyaluronate, hyaluronan.
Properties
Name and Source: Hyaluronic acid (HA, Hyaluronan, Hyaluronate) is an anionic, non-sulfated glycosaminoglycan distributed widely throughout connective, epithelial, and neural tissues. CAS# 9004-61-9. Our HA products are produced via microbial fermentation. See Documents section for details.
Appearance: Lyophilized powder. The color depends on the color of the dye and the dye labeling ratio.
Solubility: Soluble in water and the solubility data varies with MW. Higher MW results in lowered solubility. Typically solubility ranges 5-50 mg/mL, that is, 0.5%-5%. Buffered solution at pH >6 enhances solubility due to deprotonation of carboxylic acid groups and formation of carboxylate salts. HA derivatives with hydrophobic groups will lower the solubility in water, and adding alcohol or DMSO to an alcohol-water or DMSO-water mixture can dramatically improve the solubility up to 200 mg/mL (20%).
Degree of Substitution (DoS): The standard DoS is 1% by default. Product variants with special DoS other than 1% is indicated in the product name and catalog number. For example, HA-635-2% means that the DoS is 2% for this particular product.
The substitution or labeling ratio is the percentage of HA disaccharide monomers that are functionalized with the functional groups. You may calculate the number of functional groups in each HA polymer chain by 1) dividing the MW of HA by the MW of one disaccharide monomer to derive the number of disaccharide monomers in HA; 2) then multiple the number of disaccharide monomers by the labeling ratio or DoS to get the number of functional groups in each HA polymer. See Documents section for details.
References
1) Biotinylated hyaluronic acid: a new tool for probing hyaluronate-receptor interactions. Bioconjug Chem., 1994, 5(4):370-2.
2) Hyaluronic acid based scaffolds for tissue engineering, A review, Carbohydrate Polymers 2013, 92(2):1262-1279. 3) An insight on hyaluronic acid in drug targeting and drug delivery, J Drug Target, 2008, 16(2):91-107.
4) Single-molecule mechanical measurements of the hyaluronan-aggrecan bottle brush, Biomolecules, 2019, arXiv:1910.13576
Click here to view an expanded list of hundreds of publications citing Creative PEGWorks products.